Understanding, using and evaluating scientific models is part of what it means to be a scientist. That’s why we’ve added learning about models and representations to the new ‘Thinking and Working Scientifically’ strand of our revised Cambridge Primary and Lower Secondary Science programmes.
By explicitly teaching about models, you can help Cambridge learners to have a fuller understanding of the nature of science and the work of scientists. Importantly you can help them to understand that the value of a model depends on the specific understanding it is intended to convey and the context it will be used in. Therefore, all models which convey some truth are useful in some contexts, regardless of how simple or complex they are.
Understanding about models can also help your learners to avoid misconceptions. This is because learners who understand which features of a model to concentrate on, are less likely to use the model in unintended, and unhelpful, ways.
Models help scientists to understand and do science
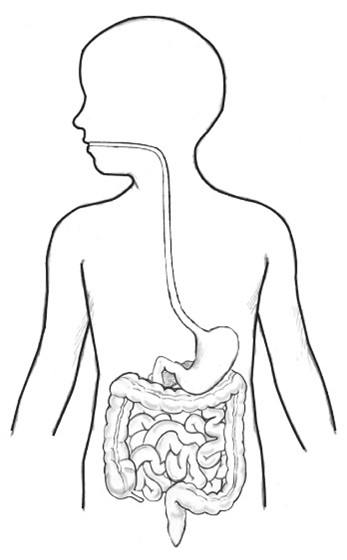
A diagram like Figure 1 (above) is a common way of checking that learners can identify and name the parts of the human digestive system. But, this is not what the inside of the human body looks like.
This is not a problem in science education because this diagram is an example of a scientific model – and scientific models do not fully represent reality. Instead, scientists use models to help explain and think about scientific ideas that are abstract or invisible. Models can focus on the very big (e.g. the Solar System), the very small (e.g. an atom) or anything in between. In the case of this diagram of the human digestive system, all other organs (including the lungs, heart and brain) are omitted to help us to focus only on the digestive system.
Building up understanding as learners progress
Cambridge Primary and Lower Secondary Science has always used models to introduce and explain scientific concepts, but now we also expect learners to understand what scientific models are and how to use them.
The learning objectives for ‘models and representations’ start in early primary with physical models and diagrams. By the end of Cambridge Primary, learners should appreciate that models are designed to show some important features of a system, but are not fully representative of a real-world situation or scientific idea. They should also know that scientists communicate their ideas in different ways and often use diagrams to represent the most important features.
In Cambridge Lower Secondary a wider range of models are considered including analogies (e.g. the Earth’s core acts like a magnet). Learners are also expected to use a wider variety of representations including symbols and formulae (e.g. CO2 for carbon dioxide or R = V/I to calculate resistance in an electrical circuit). In addition, learners start to evaluate models by describing their strengths and limitations.
An example of progression through the Cambridge Pathway: Understanding atoms
Understanding and explaining the properties of atoms has interested scientists for millennia. It underpins the work of scientists in every discipline and so we will use it as an example of the progression in thinking about, and using, models through the Cambridge Pathway.
At each stage in the history of atomic theory, the models used to describe atoms have become more complex and able to explain more phenomena. For example, Democritus (c. 460 BCE to c. 370 BCE) is famous for coining the term atomos (literally ‘indivisible’) to describe the building blocks of all matter. He thought that these atomos came in different sizes and shapes that gave different materials their different properties. Today, around 2500 years later, the standard model of fundamental particles is vastly more complicated.
This increase in complexity is also seen in how we teach learners about atomic models. We start with simple models to explain some key ideas, then we progress to more sophisticated models. Alongside this, we want our learners to progress in their understanding about scientific models. This progression can be seen in the different stages of the Cambridge Pathway:
Cambridge Primary (age 5 to 11)
Learners are introduced to the particle model to explain the properties of solids, liquids and gases. In their learning about models, they understand that the diagrams they draw of the particles are not pictures of what is ‘really there’ but are ways to highlight some important ideas (e.g. the arrangement of the particles in the different states).

Cambridge Lower Secondary (age 11 to 14)
In Lower Secondary, learners start to use more sophisticated atomic models. In Stage 8 of the curriculum they use the Rutherford model (a central nucleus surrounded by electrons, proposed 1911). Then, in Stage 9, they use the Rutherford-Bohr model (that electrons orbit the nucleus in defined shells, proposed 1913) and link this to the structure of the Periodic Table (itself another important scientific model).
In terms of their understanding about models, they consider the use of analogies (e.g. the atom is ‘like the solar system’) and use symbols such as dots and crosses to represent electrons in chemical bonds. They can also consider the strengths and limitations of the atomic models they have studied, by identifying that the limitations of one model can lead to scientists creating more sophisticated models. For example, the Rutherford model was created in response to experimental results that could not be explained by the atomic model common at the time.
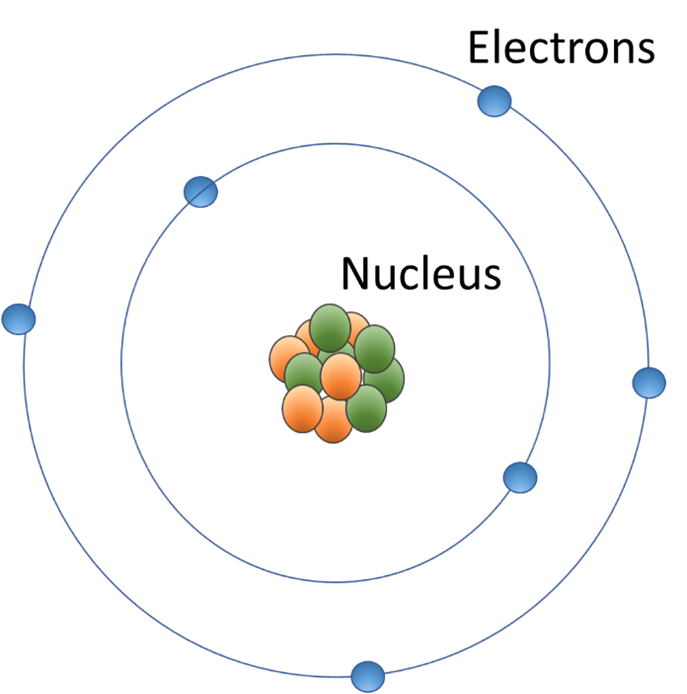
Cambridge Upper Secondary and Advanced (age 16 to 19)
Understanding the Bohr-Rutherford model in Cambridge Lower Secondary Science equips learners for the level of atomic understanding expected by Cambridge IGCSE™ Sciences. This, in turn, prepares learners for more advanced atomic models. In Cambridge International AS & A Level Chemistry, learners use a more sophisticated model for electron behaviour which includes shells, subshells and orbitals. In Cambridge International AS & A Level Physics, learners are introduced to the fundamental particles described in the standard model. These models in turn are developed further in university courses.
Take a more detailed look…
In 2020 we introduced some exciting changes to the English, Maths and Science curricula for first teaching in September 2021. The changes we made to the Science curricula included adding a new strand called ‘Thinking and Working Scientifically’ – designed to help younger learners think like scientists and prepare them for success in their future science studies. The specific teaching of scientific models supports this aim.
To find out more about our science programmes for 5 to 14 year-olds, please take a look at our Cambridge Primary and Lower Secondary web pages.